By Paul T. Finger, MD
Description
When possible, many centers offer radiation as an eye and vision-sparing alternative for patients with intraocular cancer. The two main types of radiation are plaque radiotherapy and external beam.
External beam radiation was commonly used for retinoblastoma (a childhood eye cancer), choroidal melanoma (in several centers) and for metastatic tumors (that have spread from another part of the body to the eye).
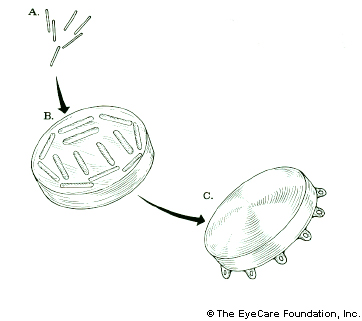
Plaque brachytherapy is the most widely used treatment for choroidal melanoma and delivers a highly concentrated radiation dose to the tumor (with relatively less radiation to surrounding healthy tissues). The radioactive plaque can also be called a “radiation implant” or “radioactive source.”
Plaque brachytherapy is typically used in one definitive treatment. The radiation sources used for brachytherapy come in the form of small “rice-sized” radioactive seeds. These seeds are attached within a gold or steel bowl called a plaque.
The dose of radiation delivered to the tumor is determined by the type, number and strength of the seeds used and length of time of the implant. The dose will also depend on the size of the tumor and its location. Make sure your eye cancer specialist has performed a comparison between various types of available eye plaques (e.g. iodine-125, palladium-103 and ruthenium-106) before surgery. Though they all can destroy the tumor, the plaque that delivers least radiation to your macula, optic nerve and fovea will offer the patient his or her best chance of keeping your vision (over time).
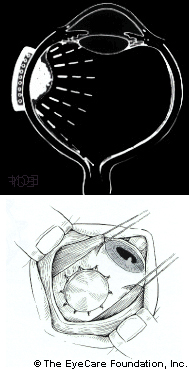
Placement of the plaque is performed in the operating room. During the procedure, the eye cancer specialists will attach the plaque to the wall of the eye, covering the base of the intraocular tumor.
A comprehensive review of radiation therapy for choroidal melanoma showed that many forms or sources of radiation have been used to treat intraocular tumors. It is reasonable to assume that given a large enough dose, all of these radiation techniques can destroy intraocular tumors. They differ in the distribution of ocular and orbital radiation dose, and their resulting radiation side effects. For example, proton radiation will affect those tissues through which it travels to get to the tumor (typically the eyelashes, eyelid, conjunctiva, cornea, lens, iris and ciliary body), whereas plaque radiation travels throught the wall of the eye (sclera and cornea) in order to reach an intraocular tumor.
Dr. Finger’s review showed that external beam therapy resulted in more reported anterior complications than plaque therapy for choroidal melanoma.
These findings, and our desire to improve radiation safety, have driven an evolution in treatment techniques.
Dr. Finger currently recommends the use of palladium-103 for plaque radiation therapy of most choroidal melanomas. Studies have shown that low-energy iodine-125 plaques have largely replaced cobalt-60 and ruthenium-106 for plaque radiation therapy.
In 1985, the Collaborative Ocular Melanoma Study chose iodine-125 ophthalmic plaque radiation therapy (iodine-125 seeds in gold ophthalmic plaques) to treat medium-sized choroidal melanomas. This decision resulted in both widespread acceptance and standardization of this technique.
Then, in 1990, palladium-103 became available for the treatment of cancer. Though there has been no randomized trial comparing palladium-103 versus iodine-125, the results of the largest studies can be seen in the detailed comparison of iodine-125, ruthenium-106 and palladium-103 plaques.
Table 5. 105Pd. vs. 125I results
| Authors | Radiation | Mean apex dose (Gy) | Follow-up (mo) | Recurrence (%) | Enucleation (%) | Metastasis (%) | Visual acuity |
|---|---|---|---|---|---|---|---|
| Packer et al. (11) | 125I results | 91 | 64 | 7.8 | 17.2 | 15.6 | 45% better or 20/100 |
| Fontanesi et al. (26) | 125I results | 79 | 46 | 2.3 | 9.7 | 5.5 | 41% better or 20/200 |
| Giblin (27) | 125I results | 97 | 60 | 7.2 | 8.8 | 6.1 | 47% better or 20/200 |
| Kreissig et al. (25) | 125I results | 70 | 60 | 10 | 0 | 15.7 | 100% “severe loss of vision” |
| Melia et al. (28) | 125I results | Variable* | 36 | NA | NA | NA | 57% better than 20/200 |
| Lommatzsch (30) | 106Ru results | 100 | 80 | 15 | 26 | 20 | NA |
| All 6 trials | Mean | 86 | 67 | 8 | 11 | 11 | |
| Finger et al. | 103Pd results | 80.5 | 55 | 4 | 6 | 6 | 73% better than 20/200 |
* Minimum 5mm “apex dose.” Abbreviation: NA = not available.
Admission to the Hospital
After admission to the hospital, you will be asked to sign a consent form (if not done previously). Your doctor may order blood tests, an EKG, x-rays or imaging scans. The placement procedure takes a short time. The radiation is painless, but sometimes movement of the eye or stitches can cause discomfort. Medication is available if you experience pain.
Activity with the Implant in Place
Most patients will be allowed to stay at home during iodine-125 or palladium-103 plaque therapy. A lead patch or lead glasses must be kept over the operated eye while the plaque is in place.
Side effects/complications
Radiation works on cells by creating free-radicals (primarily hydroxyl OH-) in humans which can directly destroy cells and by breaking cellular DNA. This means that there are some immediate tumor killing effects and other effects which will not show up until the cell tries to divide (using its broken DNA). Cells that will die when they try to divide are considered sterile and incapable of metastasis. Radiation damages both the cancerous and normal tissues. This is one reason why tumors tend to shrink slowly, and why some radiation complications appear years after treatment.
Complications can occur after radiation therapy for intraocular and orbital tumors. The incidence of these complications is related to the amount (dose) given, how fast it is given (dose-rate), the age of the patient, other synchronous diseases, and the individuals tolerance for radiation therapy. In general, larger tumors require more radiation to kill them as do some tumors which are less radiation sensitive.
Large differences exist between plaque therapy which typically deliver radiation constantly for 5-7 days, as compared to external beam therapy, which typically delivers 5 daily doses of 1200-1500 cGy in 5 minutes (each). Both require surgery.
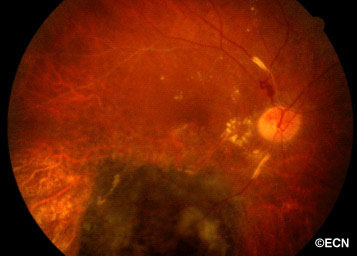 Above, this is an intraocular photograph of a patient treated with ophthalmic plaque radiation who later developed optic nerve neovascularization, intraretinal microangiopathy, chorioretinal atrophy, a ghost vessel, and perivascular sheathing. The dark tumor on the bottom of the photograph was inactive (sterilized).
Above, this is an intraocular photograph of a patient treated with ophthalmic plaque radiation who later developed optic nerve neovascularization, intraretinal microangiopathy, chorioretinal atrophy, a ghost vessel, and perivascular sheathing. The dark tumor on the bottom of the photograph was inactive (sterilized).
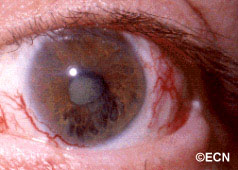
Above, this patient was treated with proton-beam irradiation. He developed radiation-related eyelash loss, dry eye, cataract, and iris neovascularization. Notice the large conjunctival blood vessels. In contrast, eyelash loss, dilated conjunctival vessels and dry eye are unusual after plaque radiation therapy.
These cases were chosen as to include most of the known complications found after radiation therapy for intraocular tumors. It is important to note that many patients do not develop these complications and benefit from the most commonly used eye and vision sparing alternative to enucleation.
Summary
The two main types of radiation are eye-plaque brachytherapy (e.g. iodine-125, ruthenium-106, palladium-103) and external beam (proton-beam). In general, external beam therapies are more likely to be associated with external (lids, cornea, iris, lens) complications and plaques appear to induce earlier radiation retinopathy with few external radiation complications.
Post treatment care
After Treatment Is Over:
You may experience some fatigue for a few weeks following surgery. You may find it helpful to plan rest periods throughout your day. You may resume your normal diet after discharge from the hospital, unless directed otherwise by your doctor.
Skin Care Around the Eye
The following guidelines will help promote comfort and healing of the area involved.
- Wash the eye lids with mild soap and lukewarm water and gently pat dry.
- Avoid extreme temperatures (e.g. hot showers, hot water bottles, heating pads, or ice bags) on the affected area.
- Avoid any friction or eyelid rubbing or scratching.
- Radiation blepharitis (eyelid inflammation) can be treated with silvadine ointment, black tea soaks, or A&D ointment.
Studies/research
Dr. Finger suggests that clinical centers perform comparative dosimetry studies of iodine-125 versus palladium-103 prior to all plaque insertions.
- Finger PT. Radiation Therapy for Choroidal Melanoma. Survey of Ophthalmology (Review Article) 42:215-32, 1997.
- Finger PT, Berson A, Szechter A. Palladium-103 Plaque Radiotherapy for Choroidal Melanoma: Results of a 7-Year Study. Ophthalmology 106:606-613, 1999
- Finger PT, Berson A, Ng T, Szechter A. Palladium-103 Plaque Radiotherapy for Choroidal Melanoma: An 11-Year Study. International Journal of Radiation Oncology Biology Physics 54;1438-1445, 2002.
- Finger PT, Chin KJ, Duvall BS for The Palladium-103 for Choroidal Melanoma Study Group. Palladium-103 Ophthalmic Plaque Radiation Therapy for Choroidal Melanoma: 400 Treated Patients. Ophthalmology 2009;116:790-6.
- Laube T, Fluhs D, Kessler C, Fiscia LE, Bornfeld N. Determination of Surgeon’s Absorbed Dose in Iodine-125 and Ruthenium-106 Ophthalmic Plaque Surgery Ophthalmology 107;107:366-369.
Related links
- Iodine-125 and palladium-103 eye plaques
- Compare the results of Iodine-125, ruthenium-106 and palladium-103 eye plaques
- Compare Proton Beam Charged Particle Radiation Therapy to Ophthalmic Plaque Irradiation
- Palladium-103 and iodine-125 plaque construction
- Tumour location affects the incidence of cataract and retinopathy after ophthalmic plaque radiation therapy.









